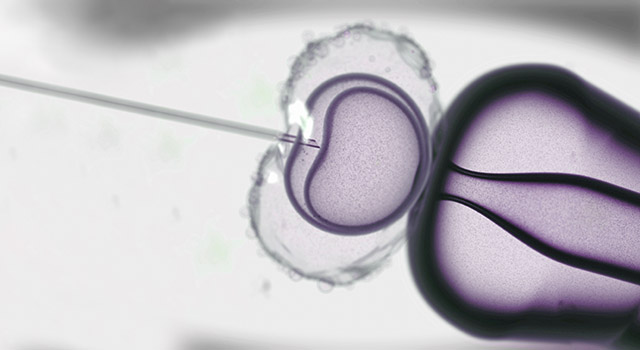
In Vitro Fertilization (IVF) is usually the best treatment option for couples whose infertility results from irreparable tubal disease, severe endometriosis, or very poor semen quality, and also offers an excellent chance for success to couples with unexplained infertility when other treatments fail. The IVF process involves a number of steps and very sophisticated medical technology, but is not difficult to understand.
IVF Overview
The key element of the IVF process involves the fertilization of the eggs “in vitro,” which simply means outside of the body. Eggs collected directly from the ovaries are combined with sperm in the laboratory to create embryos, which are then grown and nurtured in specialized incubators for three to five days before the best of the group is selected for transfer into the uterus, through the vagina and cervix.
- The ovaries are stimulated by treatment with potent injectable fertility medications, called gonadotropins, which are purified concentrates of the same hormones the body normally makes, but administered in doses greater than would normally be produced, to stimulate the growth and development of a group of follicles, each of which contains one egg. By contrast, in the natural menstrual cycle, usually only one follicle reaches maturity and only one egg is released each month.
- The response to stimulation is monitored carefully in brief office visits every one to two days. Ultrasound examinations reveal the number and size of the growing follicles, and blood estrogen measurements reflect the maturity of the follicles and the eggs within. Results are used to guide adjustments in the dose of the stimulating hormones. The stimulation phase of the IVF process usually takes eight to twelve days, similar to the length of the time between the onset of a menstrual period and ovulation (release of the egg) in the natural cycle.
- When the group of growing follicles reaches maturity, treatment with the stimulating hormones ends and a “trigger” injection is administered to complete the final stages of egg development. Approximately 36 hours later, the eggs are collected directly from the ovaries by transvaginal ultrasound-guided needle aspiration, performed under intravenous sedation. Patients are sedated to an extent that prevents them from experiencing any pain but allows them to still breathe on their own. The procedure usually takes 20-30 minutes to perform, and because the sedating medications have a very short duration of action, most patients are awake, alert and able to leave the office after about one hour.
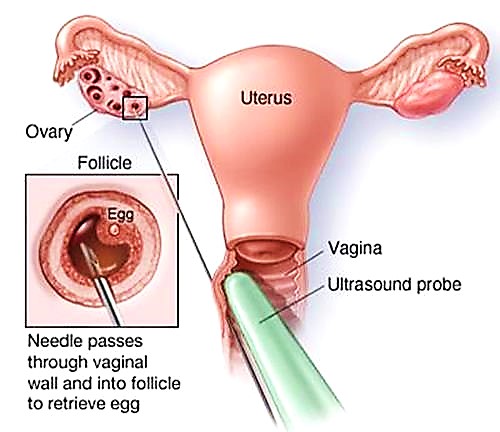
- The fluids aspirated from the ovarian follicles are examined immediately in the adjacent laboratory and the eggs within are identified and isolated by the embryologist.
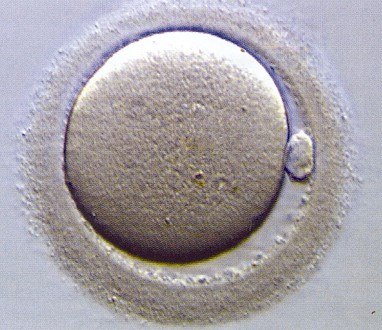 Mature human oocyte (egg)
Mature human oocyte (egg)
- That same day, the eggs are fertilized with sperm in the laboratory and then placed in highly specialized incubators that carefully control their environment. The next morning, they are examined under a microscope to determine how many in the group fertilized successfully; on average 60-80% of the eggs that were fully mature can be expected to fertilize.
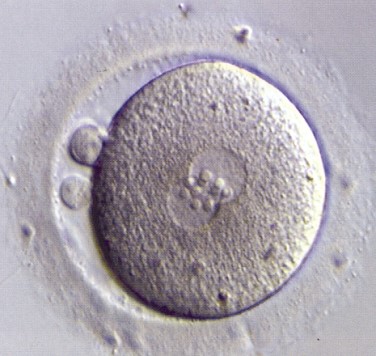 Fertilized egg (zygote)
Fertilized egg (zygote)
- The growing embryos are examined again on the third day after fertilization, at which time normally developing embryos typically contain 6-8 cells (the “cleavage stage” of development). If there are a number of high quality embryos, they will remain in the incubator for another two days. On the fifth day after fertilization, normally developing embryos reach the “blastocyst” stage of development, the same stage they are when they arrive in the uterus in a naturally conceived pregnancy. At this stage, the rapidly increasing number of cells in the embryo begins to separate into those that will become the baby (called the inner cell mass) and those that will become the placenta (called the trophectoderm).
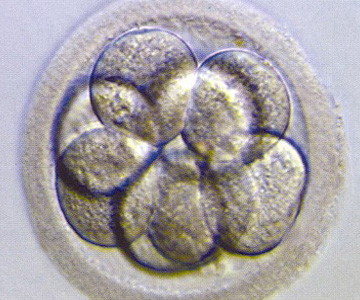 Cleavage stage embryo (Day 3)
Cleavage stage embryo (Day 3)
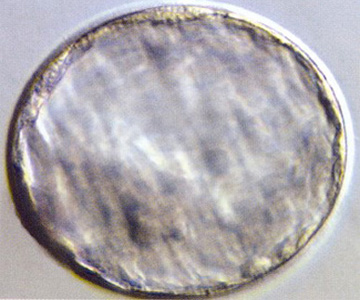 Blastocyst (Day 5)
Blastocyst (Day 5)
- On the fifth day after fertilization (sometimes earlier, on the third day), the best of the available embryos are transferred directly into the uterus, through the cervix under ultrasound guidance, in a brief outpatient procedure that does not require sedation or any other specific preparation.
Our practice adheres closely to the practice guidelines issued by the American Society for Reproductive Medicine and the Society for Assisted Reproductive Technologies (shown below), which define the maximum number of embryos recommended for transfer, according to age. In patients with a good prognosis, based primarily on age (< 35 years), history (first IVF cycle or previous IVF success) who have a number of high quality embryos, the usual best practice is to transfer the ONE best embryo to avoid the otherwise substantial risk for conceiving a multiple pregnancy.
| Age | ||||
| Prognosis |
<35 |
35-37 |
38-40 |
41-42 |
| Cleavage stage embryos | ||||
| Favorable |
1 |
1 |
= 3 |
= 4 |
| All others |
= 2 |
= 3 |
= 4 |
= 5 |
| Blastocysts | ||||
| Euploid |
1 |
1 |
1 |
1 |
| Other Favorable |
1 |
1 |
= 2 |
= 3 |
| All others |
= 2 |
= 2 |
= 3 |
= 3 |
Favorable = Any ONE of these criteria: Fresh cycle: euploid embryos, expectation of 1 or more high-quality embryos available for cryopreservation, or previous live birth after an IVF cycle. FET cycle: availability of vitrified day-5 or day-6 blastocysts, euploid embryos, 1st FET cycle, or previous live birth after an IVF cycle.
- A hormone support treatment regimen continues from the time of transfer until a pregnancy test is performed 9-11 days after embryo transfer. Patients who achieve pregnancy are followed closely, first with pregnancy hormone (hCG) measurements, and then with weekly transvaginal ultrasound examinations until approximately 8 weeks of gestation (4 weeks after the positive pregnancy test) and then transition to obstetrical care with the physician(s) of their choice.
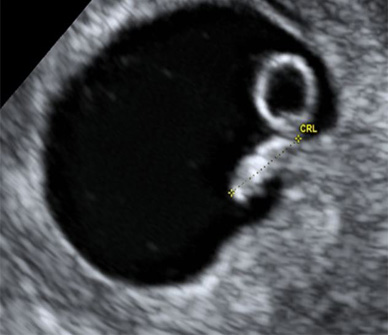 6 weeks, 3 days gestation (4.0 mm)
6 weeks, 3 days gestation (4.0 mm)
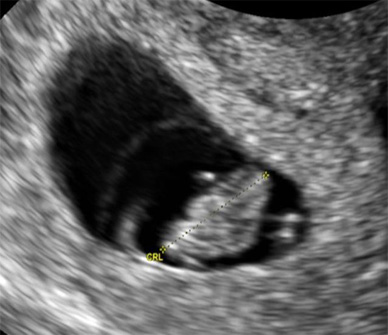 7 weeks, 5 days gestation (14.0 mm)
7 weeks, 5 days gestation (14.0 mm)
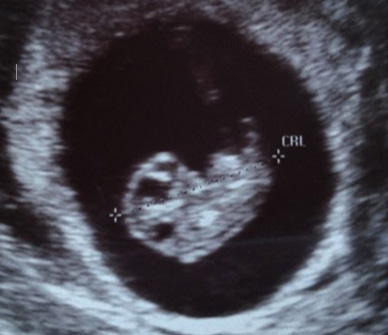 8 weeks, 3 days gestation (20.5mm)
8 weeks, 3 days gestation (20.5mm)
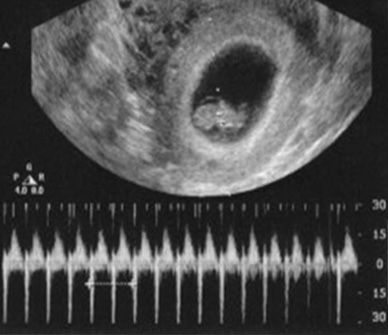 Fetal heart activity
Fetal heart activity
Success Rates
Many factors affect the likelihood for achieving success with In Vitro Fertilization, but the most important one, by far, is age.
In general, female fertility peaks between the ages of 20 and 24 and then begins to decline, but relatively little until approximately age 32, when fertility begins to decline steadily, and at a rate that increases as age increases. Unlike men, whose testicles make sperm on a continuous basis, women are born with all of the eggs they will ever have. In each menstrual cycle, a group of ovarian follicles, each of which contains one egg, begins to grow and mature, and the number in each group gradually decreases over time as the overall size of the remaining egg supply declines. Consequently, more follicles can be expected to grow in younger women than in older women, so the number of eggs that can be retrieved in an IVF cycle decreases with age.
Not every follicle that grows will yield an egg, not every egg that is retrieved will be fully mature or fertilize, not every fertilized egg will become an embryo, and not every embryo that begins to grow will continue to develop. In younger women, fewer eggs are needed to achieve success, because the quality and developmental potential of each egg is high. In older women, more eggs are required to achieve success, but at the same time, the number that can be stimulated to grow and that can be retrieved is smaller. As a result, the likelihood for achieving a successful pregnancy in each IVF cycle naturally decreases with age.


At UNC Fertility, success means patient satisfaction achieved through providing the utmost level of patient care. We hold ourselves to the highest ethical standards and it is demonstrated by having some of the best success rates in the country. Our success rates are submitted to a nationwide organization called SART, the Society for Assisted Reproductive Technology. SART is an organization dedicated to IVF and assisted reproductive technologies (ARTs). Their mission is to establish and maintain the standards for IVF and ARTs so that patients can receive the highest level of care possible. It is important to mention that not all clinics report their data to SART for numerous reasons. At UNC Fertility, we are proud to submit our success rates and to be nationally recognized by SART.
For a review of UNC Fertility’s most recent statistics*, as reported by the Society for Assisted Reproductive Technology, please visit the following website: click here
Monitoring
The response to ovarian stimulation during a cycle of In Vitro Fertilization must be monitored carefully to ensure that treatment is optimized. Monitoring includes transvaginal ultrasound examinations to observe the number and size of the developing ovarian follicles and the growth of the uterine lining (called the endometrium), and blood hormone measurements to help in assessing their maturity. In general, 4-5 monitoring visits will be required during the course of stimulation.
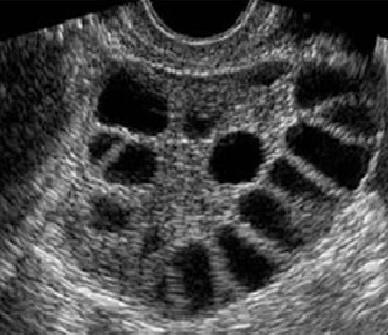 Ovary with developing follices
Ovary with developing follices
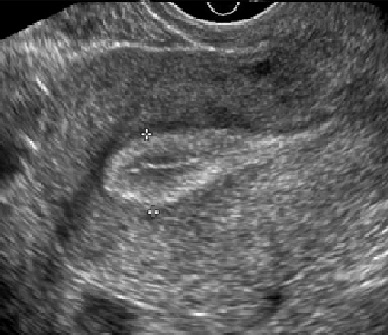 Uterine lining development
Uterine lining development
UNC Fertility is open seven days a week and monitoring procedures are performed in the early morning hours so that all results can be available by early in the afternoon. You will be contacted in the afternoon of each monitoring visit to inform you of the results, any adjustments in your treatment plan, and the day and time for your next visit.
Frozen Embryo Transfer
IVF cycles frequently yield more embryos than is appropriate to transfer to the uterus. Quality embryos that have developed normally but are not selected for transfer can be frozen for future use if the patient/couple chooses. Frozen embryos can remain in storage indefinitely and offer additional opportunities for pregnancy if the fresh embryo transfer is not successful, or when the patient/couple is ready to have a second or later child. Approximately 90% of frozen embryos can be expected to survive after thawing.
We believe an individualized treatment plan and specialized care is imperative to helping you achieve your ultimate goal of bringing home a baby. The expertise and experience of our dedicated medical team, including highly experienced embryologists, laboratory support staff and specially trained nurses contribute greatly to our success rates.
A frozen embryo transfer cycle is less complicated than a fresh IVF cycle because all of the work involved in ovarian stimulation, egg retrieval, fertilization, and embryo culture is already done. The uterus is prepared to receive frozen embryos by treatment with estrogen for approximately two weeks, to stimulate growth of the uterine lining, then adding treatment with progesterone, to mature and synchronize the development of the uterine lining with the developmental stage of the embryo(s). The embryo transfer procedure for frozen-thawed embryos is identical to that used for transfer of fresh embryos. Success rates after frozen embryo transfers generally average about two-thirds of those expected for fresh transfer cycles.
Related IVF Content
Intracytoplasmic Sperm Injection (ICSI)
ICSI helps maximize the number of fertilized eggs when there is an issue with the sperm, and in couples with unexplained infertility.
Assisted hatching involved making a small opening int eh “shell” that surrounds the embryo, increasing the chance for embryo implantation.
Blastocyst transfer involves a longer duration of embryo culture in the laboratory (5 days), which helps to identify the most viable embryos.
Preimplantation Genetic Testing
PGS is a method of testing the chromosomes in developing embryos to help identify those that are most likely to implant and becomes healthy baby.
For a consultation at our UNC Fertility Clinic in Raleigh and to discuss if IVF is right for you, contact us today to schedule an appointment. We have helped thousands of patients with their infertility issues through IVF. We see patients from all surrounding areas including Raleigh, Durham, Cary and Fayetteville.
*For a number of reasons, the most recent data for all centers are approximately two years old. According to SART, “A comparison of clinic success rates may not be meaningful because patient medical characteristics and treatment approaches may vary from clinic to clinic.”